
To explain the history of nuclear energy we could distinguish three major stages:
- Physical and chemical scientific studies of the elements.
- The development of the nuclear bomb during World War II.
- Use of nuclear energy in the civil sphere.
Physical and chemical scientific studies of the elements
The development of nuclear energy has its roots in scientific advances in physics and chemistry, which began centuries ago and culminated in the discovery of nuclear fission in the 20th century.
These studies focused on understanding the structure of matter and the phenomena related to the interaction between subatomic particles.
Atomic theory in antiquity
The idea of the atom as the fundamental unit of matter has its origins in Ancient Greece, when philosophers such as Democritus of Abdera proposed that all matter was composed of indivisible and indestructible particles called "atoms." Although their theory lacked experimental basis, it laid the first conceptual foundations for the study of matter.
The revival of atomic theory
During the 17th and 18th centuries, the idea of atoms was revitalized by the experiments of chemists such as Antoine Lavoisier, who established the Law of Conservation of Matter.
John Dalton, in the early 19th century, introduced his atomic model, based on the idea that elements are made up of identical atoms, and that chemical compounds are combinations of these atoms in defined proportions.
Subatomic discoveries
The development of scientific instruments and experimental methods allowed a more detailed understanding of atomic structure in the late 19th and early 20th centuries:
- Electron (JJ Thomson, 1897): Thomson discovered the electron and proposed the "plum pudding" model, where electrons were embedded in a "dough" of positive charge.
- Natural radioactivity (Antoine-Henri Becquerel, 1896): Becquerel discovered that certain elements spontaneously emitted radiation, a phenomenon that was later studied by Marie and Pierre Curie, who isolated the radioactive elements polonium and radium.
- Nuclear model (Ernest Rutherford, 1911): Rutherford performed experiments that demonstrated the existence of a small, dense, positively charged nucleus surrounded by electrons.
- Energy quanta (Max Planck, 1900): Planck introduced the idea that energy is not emitted continuously, but in discrete packets called "quanta", laying the foundations of quantum mechanics.
- Theory of relativity (Albert Einstein, 1905): Einstein related energy and mass with his famous equation E=mc2E = mc^2E=mc2, which would be key to understanding how nuclear fission can release enormous amounts of energy.
- Discovery of the neutron (James Chadwick, 1932): Chadwick identified the neutron, a neutral particle present in the nucleus, which facilitated the study of nuclear reactions.
Nuclear fission
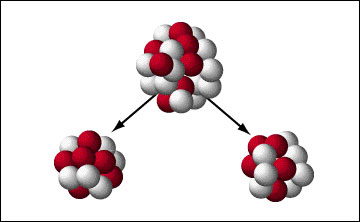 In 1938, a team of German scientists, led by Otto Hahn and Fritz Strassmann, and with theoretical contributions from Lise Meitner and Otto Frisch, discovered nuclear fission.
In 1938, a team of German scientists, led by Otto Hahn and Fritz Strassmann, and with theoretical contributions from Lise Meitner and Otto Frisch, discovered nuclear fission.
This phenomenon occurs when the nucleus of a heavy atom, such as uranium-235, splits into two lighter nuclei when bombarded with neutrons, releasing an enormous amount of energy and more neutrons, which can trigger a chain reaction.
This discovery marked the turning point between basic scientific studies and the development of practical applications of nuclear energy, both military and civilian.
Discovery of nuclear fission and military development
The discovery of nuclear fission (1938)
In December 1938, German chemists Otto Hahn and Fritz Strassmann discovered that bombarding uranium atoms with neutrons caused them to split into two lighter nuclei, such as barium and krypton. This process, known as nuclear fission , released an enormous amount of energy.
The theoretical interpretation of the phenomenon was carried out by Lise Meitner and her nephew Otto Frisch. They explained that, when dividing, the nucleus released a fraction of the original mass as energy, in line with Einstein's equation E=mc 2. Frisch coined the term "fission" by comparing it to the biological process of cell division.
This discovery revolutionized nuclear physics and marked the beginning of a new era in energy and weapons production.
Historical context and military application
The discovery of nuclear fission occurred in a context of political tensions and global conflicts that led to the Second World War (1939-1945). Scientists quickly understood that nuclear fission could be used to create weapons of unprecedented power.
In 1939, Albert Einstein and Leó Szilárd wrote a letter to U.S. President Franklin D. Roosevelt, warning of Germany's potential to develop a nuclear bomb and recommending that the United States initiate a similar program.
The Manhattan Project (1942-1946)
The United States, with support from the United Kingdom and Canada, launched the Manhattan Project , a scientific-military effort aimed at developing nuclear weapons before Germany could do so. This project became one of the most ambitious and secretive programs in history.
- Organization: The project was directed by General Leslie Groves, while physicist J. Robert Oppenheimer supervised scientific research at the Los Alamos, New Mexico laboratory.
- International participation: Prominent scientists such as Enrico Fermi, Niels Bohr, Richard Feynman and Edward Teller participated.
- Materials production: Facilities were built to produce uranium-235 and plutonium-239, the key elements for nuclear weapons. The Oak Ridge, Tennessee, and Hanford, Washington, plants played key roles in this process.
First nuclear test
On July 16, 1945, the first nuclear test, known as "Trinity" , was carried out in the Alamogordo Desert of New Mexico. A plutonium-based bomb design was used. The explosion produced an energy equivalent to 21 kilotons of TNT, creating a mushroom cloud and marking the beginning of the nuclear age.
Hiroshima and Nagasaki bombings
On August 6, 1945, the United States dropped the "Little Boy" atomic bomb on Hiroshima, Japan.
This uranium-235-based bomb detonated with a yield of 15 kilotons of TNT, leveling much of the city and causing the immediate deaths of approximately 70,000 people. Thousands more died in the days and years that followed from burns, wounds, and radiation exposure.
Three days later, on August 9, a second bomb, "Fat Man" , was dropped on Nagasaki. This one, based on plutonium-239, produced a 21 kiloton explosion, instantly killing about 40,000 people and leaving many others seriously affected.
These devastating actions precipitated Japan's unconditional surrender on August 15, 1945, ending World War II.
However, the consequences of the atomic bombings were profound and far-reaching. On the one hand, they established the United States as a military superpower, marking the beginning of the Cold War by triggering an arms race with the Soviet Union. On the other hand, they generated intense ethical and political debates about the use of nuclear weapons, due to the human suffering and implications for humanity.
These events marked a turning point in the world's military and technological history.
The discovery of nuclear fission, combined with the Manhattan Project, marked a turning point in history, with profound implications for both warfare and the development of civilian nuclear power in the decades that followed.


After the explosion over Hiroshima, Einstein commented: "I should burn the fingers with which I wrote that first letter to Roosevelt."
Civil applications and international regulation
The shift towards peaceful applications
Following World War II, the perception of nuclear energy began to transform, focusing on its civilian applications and its ability to benefit humanity.
In 1953, U.S. President Dwight D. Eisenhower introduced the Atoms for Peace program . This program promoted international cooperation in the peaceful use of nuclear energy, opening the door to broader development in fields such as electricity generation, medicine, and scientific research.
Development of nuclear power plants
The use of nuclear energy as a source of electricity took a significant step forward in 1954, when the Soviet Union opened the world's first commercial nuclear power plant in Obninsk. This milestone marked the beginning of a new era in power generation.
During the 1960s and 1970s, many countries initiated nuclear programs to meet growing energy demands.
Currently, more than 450 nuclear reactors operate around the world, providing a significant portion of global electricity. These reactors stand out for their ability to generate energy efficiently and with low carbon emissions.
However, this development also poses significant challenges, such as the management of radioactive waste and the need to ensure high safety standards at nuclear plants.
International regulation for safe use
To oversee the safe and peaceful use of nuclear energy, international bodies and initiatives have been established. In 1957, the International Atomic Energy Agency (IAEA) was established , with the main objective of promoting the safe use of nuclear energy and establishing global safety standards.
On the other hand, the Nuclear Non-Proliferation Treaty (NPT) came into force in 1970. This treaty seeks to prevent the proliferation of nuclear weapons, promote disarmament and ensure that all countries can access nuclear technology for peaceful purposes.
These international regulations represent a coordinated effort to maximize the benefits of nuclear energy while mitigating its risks, balancing its potential for global development with the responsibility to avoid its potential dangers.